Why atoms are unstable in classical physics
November 24, 2016 24 Comments
Why are atoms unstable in classical physics?
Summary: The nucleus of an atom is a positive charge, an electron is a negative charge. Positive charges attract negative charges as a result of electric forces between them. This attraction causes electrons to orbit nuclei according to classical physics. But electron orbits are unstable in classical physics. Charges exert forces on other charges and those forces are described by fields. The field tells you what the force on a charge would be if the charge was at a particular position. An electric field describes the forces on a static charge from another static charge. A magnetic field is basically the electric field produced by a charge moving at constant speed. Electric and magnetic fields are so closely related that they are often described in terms of a single field called the electromagnetic field. An accelerating charge produces waves in the electromagnetic field since the field has to transition between its electric and magnetic components and vice versa. Those waves carry away energy from the accelerating charge. An orbiting electron is an accelerating charge, so this mechanism causes it to lose energy and spiral into the nucleus. This happens in a very short time, around s.
An electron accelerates when it is orbiting
The nucleus of an atom is a positive charge, an electron is a negative charge. Positive charges attract negative charges as a result of electric forces between them. This attraction causes electrons to orbit nuclei according to classical physics.
An electron orbiting a nucleus is changing direction at every point on its orbit, as illustrated in Figure 1:

Figure 1 – An object in a circular orbit is accelerating at every point on the orbit. The same would be true for an elliptical orbit.
As a result, its velocity is changing: it is accelerating. An accelerating charge radiates, so the electron loses energy by radiating. Since the electron is losing energy it falls into the nucleus. You might think that the electron could lose energy without falling into the nucleus. A car loses energy in the form of stored fuel when you drive it, but it doesn’t just fall into a ditch as a result. But an electron doesn’t store fuel. All of its energy is tied up in its motion since it has nowhere else to store the energy. So when the electron loses energy it loses speed and it falls toward the nucleus as a result.
Why does an accelerating charge radiate? To understand that, we have to know some stuff about charges in motion, fields and forces.
Forces and fields
A force is any influence on a physical system that can cause it to accelerate or decelerate: Newton’s first law of motion. The size of the force on an object is the mass of the object times the acceleration produced by the force: Newton’s second law of motion. If physical system 1 exerts a force on system 2, then system 2 exerts the same amount of force on system 1: Newton’s third law of motion.
The nucleus exerts a force on the electron before it comes in contact with the electron. As a result, at each point in space there is a vector that describes the forces an electron would experience if it was at that point. A particle with a different charge would experience a different force. The electric force depends linearly on charge. So it is useful to define a vector at each point in space that doesn’t depend on the size of the charge of the electron that can be used to help describe the forces on a particle without giving a charge in advance. The electric field is a vector valued quantity defined at every point that gives the force applied on a charged particle at that point due to other static charges.
If you had a charged object much larger than an electron, the field might be different at different parts of the object. As a result different parts of the object would experience different amounts of force. So larger objects introduce additional complications to trying to understand what’s going on. It’s more useful for this discussion to consider objects that are so small that the change in the field over those objects is so small it produces negligible internal forces: such objects are called point charges.
You can think of the electric field of a particle in terms of field lines. Field lines are a picture representation of the field. The field lines have arrows on them. The arrows point in the direction a positive charge would move in that field. More lines per unit area means more force.
A point charge will have field lines going out evenly in every direction. So a point positive charge would look like this (figure 2):

Figure 2 – Electric field lines of a stationary charge.
A positive charge would move toward a negative charge. So a point negative charge would have a similar diagram with the lines pointing toward it.
Moving charges
What about a moving charge? The field lines in the direction of the charge’s motion would shrink in that direction. Why objects shrink in the direction of their motion is explained by special relativity. As a result, the field lines get shorter parallel to the charge’s motion, but not at right angles to it. So all the field lines that are not perfectly parallel to the particle’s direction of motion change their slope so they are pointing at a steeper angle to the direction of motion (figure 3):
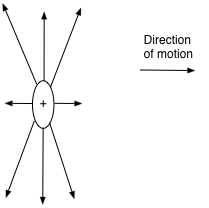
Figure 3 – Field lines of a charge moving at a constant velocity.
So it’s like there is more charge at right angles to the charge’s direction of motion. So the field at right angles to the charge gets larger.
There is one more issue to understand before I can explain why accelerating charges radiate: the relationship between electricity and magnetism. Magnets exert a force on charged particles like electrons. This effect was used in televisions until about 5-10 years ago. The inside of the television screen was coated with pixels that included materials that would glow different colours when bombarded with electrons. The TV would produce a beam of electrons and move it back and forth across that screen to produce images in quick succession. The path of the electrons was controlled by a magnet that deflected the beam to the appropriate place on the screen. Magnetic forces on a particle depend on the particle’s velocity and charge. At any given point in space, there is a vector that could be combined with the charge and velocity of a point charge at that point to give the magnetic force on that point charge: this vector is called the magnetic field.
Changing electric fields give rise to magnetic fields and vice versa. You can see this effect in action in some scrapyards where people use cranes with electromagnets. These cranes have a lump of iron with wires wrapped around it in a circular pattern, like so:
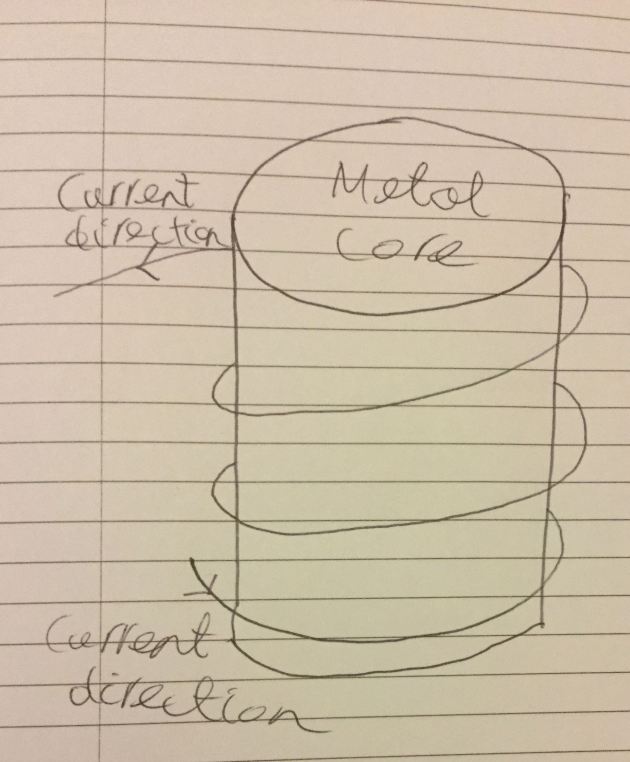
This arrangement produces a magnetic field running through the curves of the coil. How do the moving charges in the wire produce a magnetic field?
Consider a wire with current flowing through it. The electrons in the wire are moving and the nuclei of the atoms in the wire are not. As noted with the moving charge in the picture above this gives the electrons a larger field at right angles to their motion. That field looks like a magnetic field to a charge outside the wire. The magnetic field lines are at right angles to the line between the wire and the point where the field is being calculated. As a result, there is a field inside the coil pointing through the coil. So magnetic fields are a result of moving charges.
Whether a charge is moving is just a matter of your velocity relative to the charge. So an electric field is also the same as a moving magnetic field. Since electric and magnetic fields are so closely related they are usually considered to be aspects of a single field called the electromagnetic field.
You might be thinking that permanent magnets don’t need to have electricity running through them to work. In permanent magnets, the magnetic field is produced by spinning electrons whose spins are aligned with one another. So the magnetic field in such magnets is produced by the motion of charges.
Accelerating charges
We can now get back to explaining why accelerating charges radiate. An electromagnetic wave is a pattern of changes in electric and magnetic fields that can move. So if an accelerating charge generates changing electric and magnetic fields, then it radiates.
The laws of physics don’t change at different speeds: an increase from 0 speed to 5, or 5000 speed to 5100 follow the same laws. So you can understand what’s going on by considering the situation where the charge starts stationary and then starts moving.
The charge starts out with field lines like those in figure 1, then the lines change to resemble the field lines in figure 2. This happens at a finite speed so the field lines further from the charge look like those in figure 1. The lines closer to the charge look like those in figure 2. Between those two sets of lines there has to be a transition in which the lines change, as illustrated in figure 4:

Figure 4 – An illustration of the transition in field lines in an accelerating charge.
The circle is just there to illustrate the spherical symmetry of the field lines further out from the charge. This transition produces changing electromagnetic fields that spread out over time, i.e. – radiation. More generally, an accelerating charge makes components of the field transition from being electric to being magnetic and vice versa. These changes produce patterns in the electromagnetic field that move away from the accelerating charge: the charge radiates.
Since an electron is an accelerating charge, it radiates. This radiation leads to the electron losing energy and falling into the nucleus of an atom in classical physics. As a result, atoms are unstable in classical physics. Since electromagnetic fields are strong, the electron’s orbit would decay quickly in a time of the order s.

> The nucleus exerts a force on the electron before it comes in contact with the electron.
why and how? should i just take this as a given kind of like gravity but a different force besides gravity?
The electric force is kinda like gravity but different in some respects.
if the electron is accelerating because it’s being pulled on by electric-gravity, why should that make it lose charge anymore than a satellite in a stable orbit around the Earth? the energy for the acceleration comes from electric-gravity, not from its own speed, in the same way the acceleration for an orbiting satellite comes from gravity not from its own speed. (i take this to be wrong, but i don’t know why.)
The electron’s energy has two components: kinetic and potential energy. The kinetic energy is the energy is has by virtue of its motion. The potential energy is the energy it has as a result of its attraction to the nucleus.
The closer the electron is to the nucleus the stronger the nucleus pulls and so the larger the magnitude of the potential energy. But that doesn’t tell you anything about the sign of the energy: it could be large and negative or large and positive closer to the nucleus.
Suppose you want to move the electron farther from the nucleus and keep it going at the same speed. Then you have to give the electron energy. So at the same speed it has more energy farther from the nucleus. But the magnitude of the potential energy is smaller farther from the nucleus, so the potential energy must be negative cuz
kinetic energy + smaller magnitude potential energy > kinetic energy + larger magnitude potential energy
only if the potential energy is negative.
So the electron is losing energy as it moves toward the nucleus since its negative potential energy is larger closer to the nucleus. It gets rid of that energy by radiating.
i don’t get it. how does this differ from a point-particle rock orbiting an idealized spherical earth? it stays the same distance away the whole time.
A rock orbiting a spherical idealised Earth doesn’t stay the same distance away. Its orbit just decays a lot slower than the orbit of an electron cuz electromagnetic forces are stronger. A small fridge magnet can hold a pin up against the gravitational force of the whole Earth.
> A rock orbiting a spherical idealised Earth doesn’t stay the same distance away.
oh i thought it did. why doesn’t it? it’s in a vacuum with no friction to slow it down.
and if the problem is orbits decaying very very slowly (e.g. over trillions of years), that doesn’t ruin classical physics, it just puts a time limit on atoms that we haven’t reached yet. so to make your case you better say something about how quickly atoms would fall apart.
The explanation for why a rock orbiting the Earth loses energy is similar to the explanation I gave for why an electron’s orbit decays. But the explanation is a bit more compliated because gravity is a bit more complicated.
The lifetime of a hydorgen atom in classical physics can be calculated and it is of the order 10^(-11)s. Cuz electromagnetic forces are a lot stronger they die away faster so orbits have to be closer, and the orbits also decay faster cuz the attraction is stronger.
where’s the lost energy going?
The lost energy is radiated as light.
a rock orbiting the earth radiates light, too?
> a rock orbiting the earth radiates light, too?
in classical physics, QM, or both?
In classical physics and in QM if an electron is going to lose energy and move to a lower state in the atom it will emit light. A rock orbiting the Earth will emit gravitational waves instead of light if its orbit is decaying.
what are gravitational waves?
why does acceleration cause energy loss? the post could really use section headings. cuz ur like gonna answer this but then u do several asides. it they were clearly labelled and then there was a clear full description of the overall point (making the argument you wanted to make originally, but had to do the asides first) it’d help.
There is a gravitational field, which is similar to the electromagnetic field but with different equations of motion. Gravitational waves are waves in the gravitational field.
I’ve put in some headings and a brief description of the asides.
summarizing:
the nucleus exerts a force on the electron. (kinda like gravity but a different force. is this an electrical or magnetic force, or something else?)
when charges move it changes their fields (shorter field lines in the direction of motion. and shorter backwards for some reason judging from the picture?)
“Whether a charge is moving is just a matter of your velocity relative to the charge.” so if you run at a stationary charge, it’ll affect the field for you i guess.
electrical and magnetic fields are related.
“An electromagnetic wave is a pattern of changes in electric and magnetic fields that can move. So if an accelerating charge generates changing electric and magnetic fields, then it radiates.” but why?
it’d help if the article provided an outline of what your argument is instead of me trying to reconstruct one. i went through in order and picked out some key points (i think) and didn’t end up with a clear ordered summary of what your point is.
I have summarised the argument and made a few adjustments to the argument for clarity.
the summary is a big help, i think it’s better than the rest.
> But electron orbits are unstable in classical physics.
i think you should say that ALL orbits are unstable in classical physics, including electron orbits. is that right? but some decay very slowly, whereas electron orbits decay very fast as you mention later.
so how do atoms work in quantum mechanics?
That will be another post.
Pingback: Why are atoms stable in quantum mechanics? | Conjectures and Refutations