Why are atoms stable in quantum mechanics?
November 30, 2016 3 Comments
In a previous post I explained why atoms are unable in classical physics. The post is about why atoms are stable in quantum mechanics.
Summary Atoms in quantum mechanics don’t suffer from the same radiation problem as atoms in classical mechanics. A quantum system exists in many instances that can interfere with one another on a small scale. As a result, on an atomic scale an electron doesn’t have a trajectory and so it can’t be said to accelerate and it doesn’t radiate. In addition, when the probability of finding an electron is highly peaked at a particular location, quantum mechanics makes the instances spread out. The potential produced by the nucleus pulls the electron instances toward the nucleus. Atoms can be stable because the spreading out produced by quantum mechanics and the attraction produced by the potential balance out.
In classical mechanics, an electron’s orbit around an atom is unstable because it emits the energy it would need to stay in orbit as light. And the electron does this because it is accelerating. To be able to say the electron is accelerating, it has to have a trajectory – a line it travels along. Then if the line changes direction or the electron speeds up along the line you can say it is accelerating. In quantum mechanics, systems sometimes don’t have trajectories.
Absence of microscopic trajectories in quantum mechanics
In quantum mechanics, particles are described very differently from how they are described in classical mechanics. Particles are more complicated than they look. Each particle exists as multiple instances. these instances are copies in the sense that they all obey the same rules. They are instances of a specific particle in the sense that they only interact with other instances of that particle. Sometimes two instances of a particle are different: they have different locations or different momentum or different values of some other measurable quantity. Sometimes these instances are all fungible – there is literally no detectable physical difference between them. Two instances of the same particle can become different and then become fungible again in a way that depends on what happened to the different versions of the particle: this process is called quantum interference.
Now suppose you have an electron in empty space near some point Pstart. Consider a point Pfinal that some instances of the electron will reach later. How does those instances get there? First instances of the electron spread out from Pstart in all directions. Some instances go to points intermediate between Pstart and Pfinal: P1 and P2. Then some instances of the electron spread out from P1 and P2 in all directions. Some of those instances end up at Pfinal. Figure 1 shows this process with the little domes over the intermediate points indicating the instances moving in all possible directions. There is no explanation of how the electron moves that refers to just one trajectory. And none of the instances individually change direction either. At each point there is some instance coming in from any given direction and another instance leaving in the same direction. And all of the instances of the electron at a given point are fungible so you can’t tell whether the one that left in a given direction came in from that direction or not. So there is no trajectory and no acceleration.
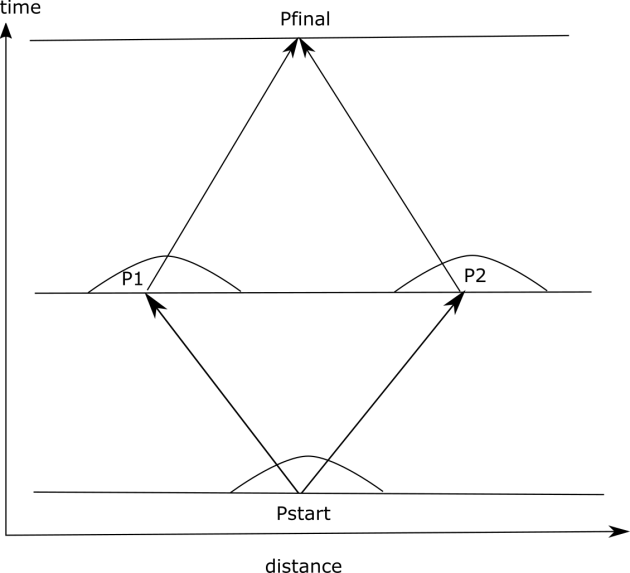
Figure 1 Instances of the electron become different and then come back together.
Now to deal with some objections you might have.
You may be thinking that people can measure where things are and this seems incompatible with there being lots of instances of the electron in different places. Quantum mechanics deals with this problem in the following way. When you do a measurement, the instances of the electron are divided up into sets. When you see some particular outcome of the measurement, the result means something like ‘this electron is within 5mm and 7mm of the corner of your desk.’ There are multiple sets of instances of the electron that give different measurement results like ‘this electron is within 0mm and 5mm of the corner of your desk’ or whatever. When you do the measurement, your instances and the instances of the measuring instrument are also divided into sets. Each of those sets acts as a record of some particular measurement result. For example, if you are detecting the electron with an instrument with a dial, there is a set of instances for each distinguishable position of the dial.
Why don’t you see multiple instances of yourself interfering in everyday life? Multiple instances of you do interfere in everyday life. They just interfere on a very small scale because it is difficult to arrange interference on a large scale. The reason it is difficult to arrange interference on a large scale is that large differences between instances can be recorded by measuring instruments and other interactions, e.g. – air molecules and light bouncing off your body. That measurement process changes the recorded instances. The only way to undo the change so the instances can become fungible again is to undo the transfer of information about the differences. You would have to track down all the light and air molecules and so on and arrange to exactly undo their interaction with you. This cannot be done with current technology so you don’t undergo quantum interference. As a result, the different instances of you don’t interfere with one another. The different instances of the objects you see around you don’t interfere with each other either. Rather, the instances form independent layers where each layer approximately obeys the laws of classical physics: parallel universes. For more explanation of quantum mechanics see The Fabric of Reality by David Deutsch, especially chapter 2, for more on quantum mechanics and fungibility see The Beginning of Infinity by David Deutsch, Chapter 11 and my post on fungibility.
The electron can have something that looks a bit like a trajectory. The electron can have more instances in some places than in others. The number of instances at different positions can be represented by a curve, like this (Figure 2):

Figure 2 A graph of number of instances with distance along some line for an electron.
If you look at a section of the curve, and find the area of the curve under that section, that tells you the probability of finding the electron in that region. In Figure 3, there is a higher probability of finding the electron in the red region since it has a higher area, so the probability of finding the electron between the two red lines is larger than the probability of finding it between the two green lines:

Figure 3 A graph of the area under the curve in two different regions of the curve.
I said that there is a number of instances, but that number is continuous and the only way to know anything about it is by calculating or measuring probabilities.
If you look at the electron on a wide enough section of the curve, then the probability of finding the electron there will be close to 1. The curve changes continuously over time so the curve could move so the peak is in different places and that could look a bit like a trajectory:

Figure 4 The curve for the electron moves around, and so the region where there is a large probability of finding the electron moves around. This is the closest thing to a trajectory in quantum mechanics.
For electrons on a large enough scale, and for large objects like a person or car, the trajectory approximation is very accurate. Things move by lumps of high probability moving from one place to another. But the scale of a single atom is small enough that the trajectory approximation doesn’t work.
Stability of atoms
The absence of trajectories by itself doesn’t explain the stability of atoms. It just explains why the problem of radiating accelerating charges doesn’t occur. To understand why atoms are are stable, let’s go back to the electron. To understand the next bit we have to know a little about how the number of instances curve changes over time. The simple version goes a bit like this:
the rate of change of the curve over time = -(curvature of the curve + the potential the electron is in).
The rate of change of the curve near a point is its slope. If the curve is very curvy, then the slope changes a lot. So the curvature is the rate of change of the rate of change of the curve. Figure 5 illustrates this with some lines near the curvy bit illustrating large change of slope, and in less curvy bit representing less change of slope.

Figure 5 The blue lines change gradient a lot over a small region, so that region has high curvature. The green lines don’t change gradient much and so the region with the green lines doesn’t have much curvature.
The rate of change of the curve over time = -curvature, so near a high peak the curvature is high and the curve gets flatter over time because it decreases at that point. Away from the peak the curvature is smaller and so the curve tends to get flatter more slowly over time. So the curvature term tends to flatten out the curve.
What about the potential? The potential is negative, as explained in the comments on the previous post. So the curve tends to get larger where the potential is large: near the nucleus. The electron can be a stable state that doesn’t change much over time if the flattening caused by the curvature term and the peaking cause by the potential match one another. In this interaction, the electron and proton are recording one another’s position, so their instances are divided up so that the electron and proton stick together.
That’s why atoms are stable in quantum physics.